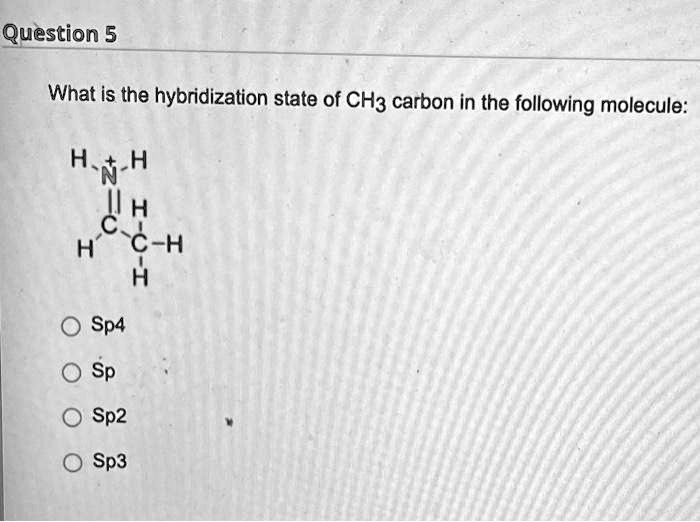
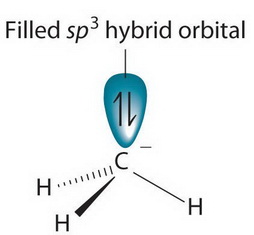
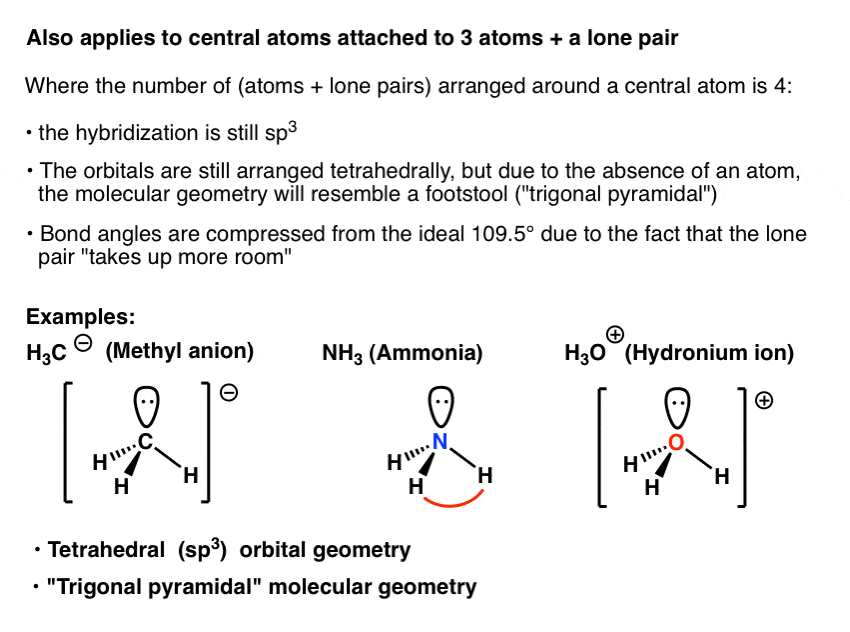
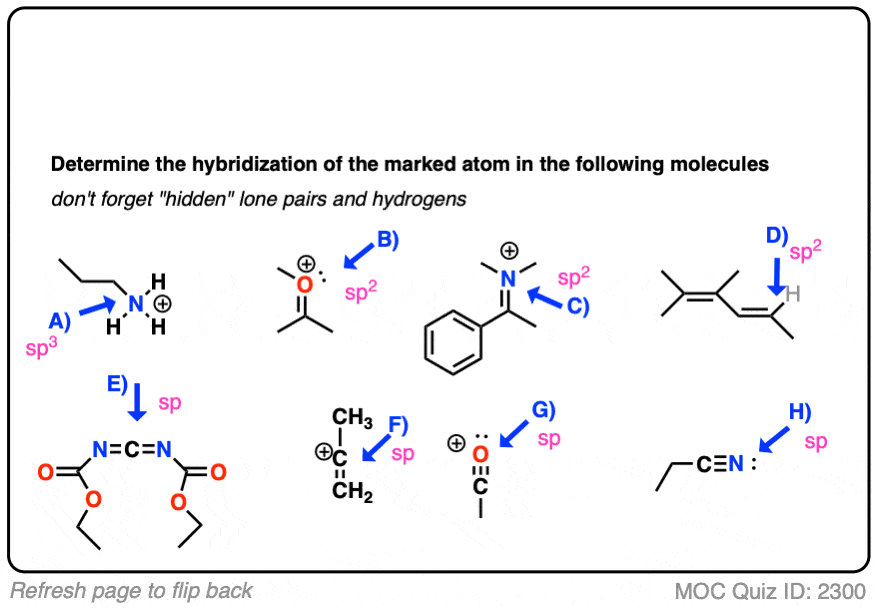
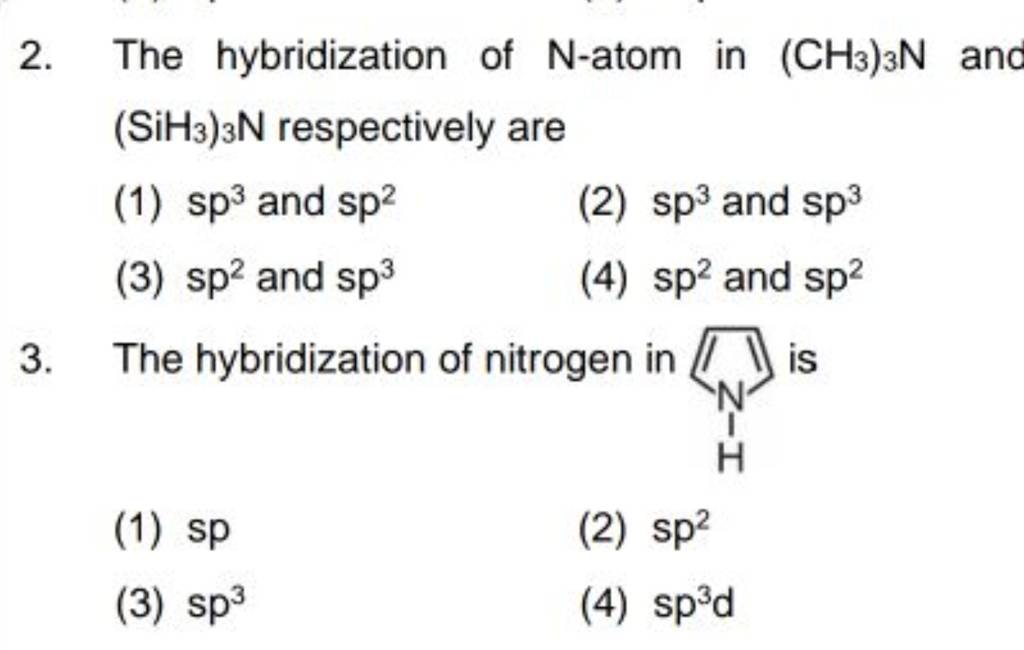
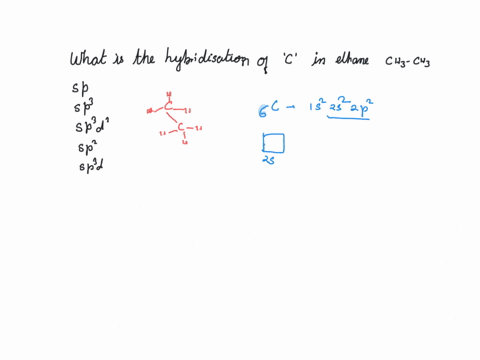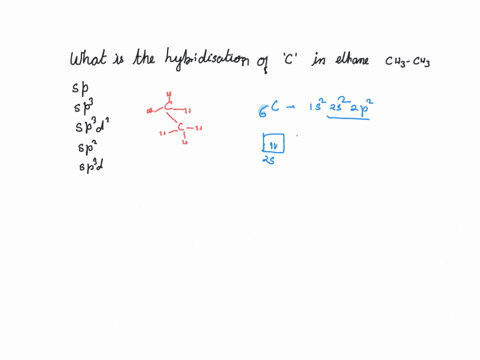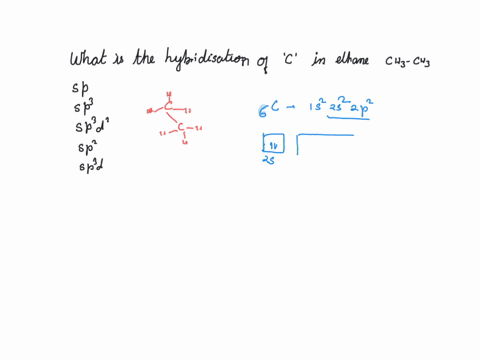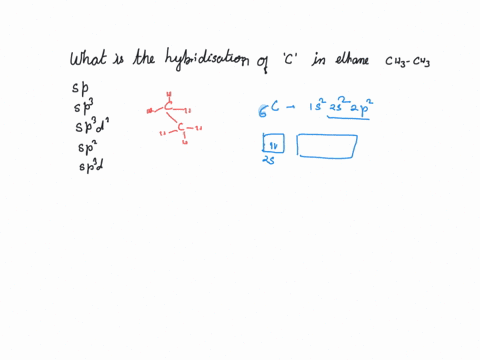CH3+and CH3- are two highly reactive carbon species. What is the predicted hybridization and geometry around each carbon atom? | Homework.Study.com

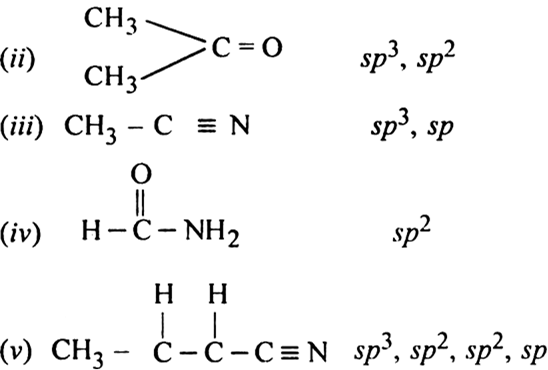
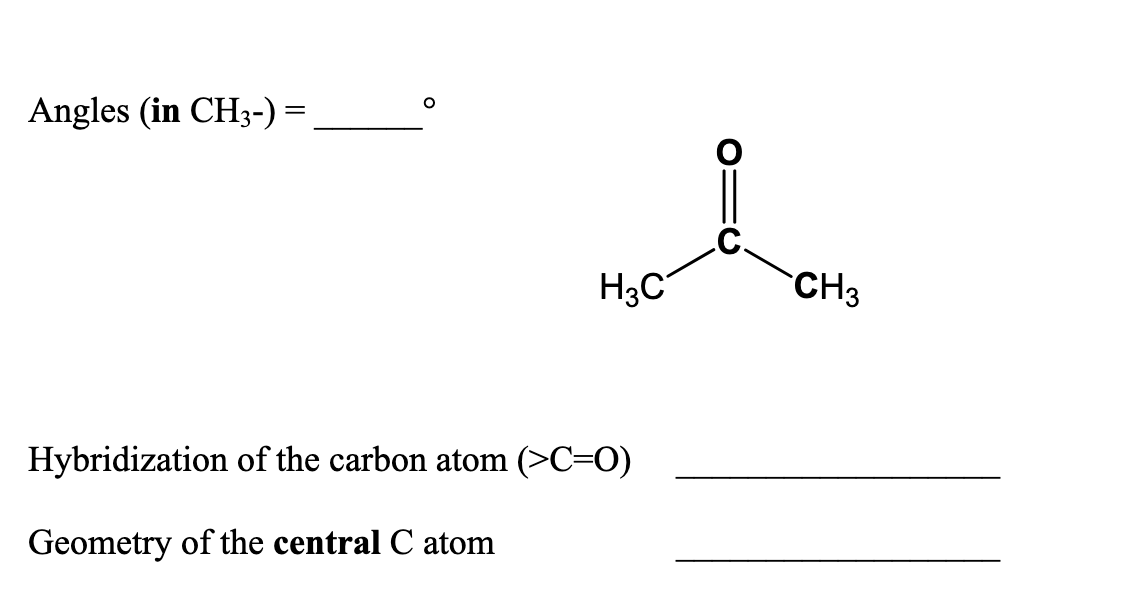
Doubt: What is the type of hybridisation of each carbon in the following compounds? (CH3)2CO , HCONH2 Chapter: Organic Chemistry - Some Basic Principles And Techniques - Subject: Chemistry - Course: NEET

Hybridization of C2 and C3 of H3C andndash; CH = C = CH andndash; CH3 area)Sp, Sp3b)Sp2, Spc)Sp2, Sp2d)Sp, SpCorrect answer is option 'B'. Can you explain this answer? - EduRev JEE